HRPP Training Requirements
Human Subject Protection Training (CITI)
All investigators and research study personnel are required to complete training regarding the protection of human subjects prior to beginning any human subject research-related activities. Initial study submissions will be halted until the required training for all study personnel has been completed and documented in the Cayuse system. This training will be verified prior to access to Cayuse Human Ethics module being granted, and MUST be completed and documented with the IRB at least every three years.
The web-based training course required by TTUHSC HRPP is the “Biomedical Investigator Course", accessed through the Collaborative Institutional Training Initiative (CITI) Program. The course takes approximately 2-4 hours to complete. TTUHSC HRPP requires
an overall score of at least 85% in order to successfully complete the course. Upon successful completion of the
course, you will be able to download a certificate of completion (for your records).
CITI Account Access
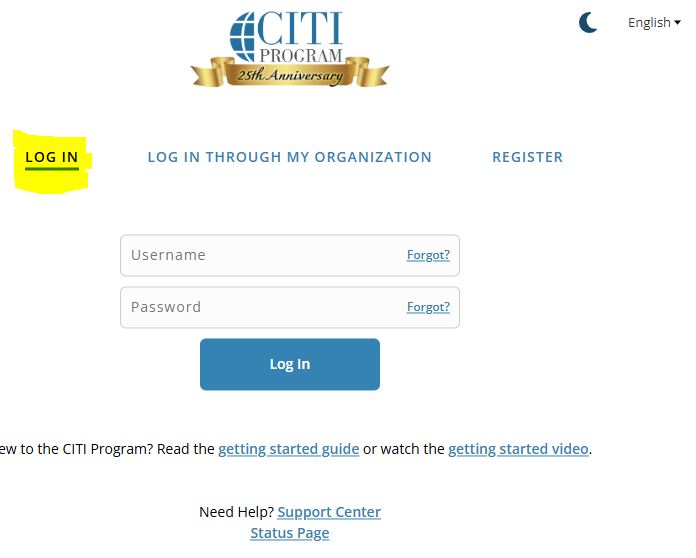
- Go to CITI login page - NOTE: If you have forgotten your Username and/or Password, use the ‘Forgot?’ option provided by CITI. Do NOT set up a new account.
- Once logged in, the page should default to the ‘My Courses’ tab. Under ‘Institutional Courses’, click on the ‘View Courses’ button for ‘Texas Tech University Health Sciences Center’. (Refer to the screenshot below)
 |
- The required course you are looking for is the “Biomedical Investigator Course”. Scroll down the page until you find this course title. NOTE: There are NO other CITI courses required by the TTUHSC HRPP.
- If the “Biomedical Investigator Course” course is not listed, you can add it (refer to the section for 'Adding / Removing a course' (under CITI Additional Information below).
- Click on the ‘Start Now’ (or ‘Continue Course’) button for the course. You will need to complete each module before moving on to the next one.
 |
TTUHSC HRPP requires an overall score of at least 85% in order to successfully complete the course. Once you have completed the course, you can go to the ‘My Records’ tab in your CITI account to access the completion report/certificate.
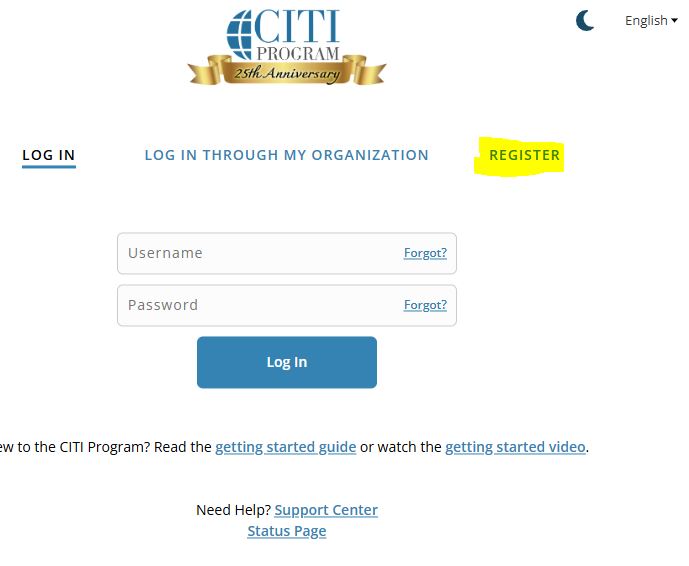
- Go to CITI login page
- Click on ‘Register’.
- On the next page, click on ‘Select Your Organization Affiliation’ and type in 'Texas Tech University Health Sciences Center'.
 |
 |
- Continue setting up your account by following the Registration steps #1-7 below.
- Steps 1-6: Answer the questions regarding ‘Personal Information’.
NOTE: Register with your TTUHSC or TTU email address unless you do not have either (i.e. you are an Outside Collaborator).
NOTE: If you are interested in CEU credit, make sure you answer the questions in Step 5 accordingly. - Step 7: It is very important that you answer these questions correctly, as they determine
which course(s) you will receive access to. Please answer as instructed below:
-
-
- Question #1 – Select ‘Biomedical Investigator Course: Research Investigators, Research Personnel, and IRB members’.
- All other questions can be skipped (unless you have been asked to complete another course by your department).
-
-
- Steps 1-6: Answer the questions regarding ‘Personal Information’.
- When you are done adding courses, click ‘Complete Registration’.
The "Biomedical Investigator Course" (and any others you have selected) will now show in your CITI account under the ‘My Courses’ tab. Click on the ‘Start Now’ button for the course. You will need to complete each module before moving on to the next one.
TTUHSC HRPP requires an overall score of at least 85% in order to successfully complete the course. Once you have completed the course, you can go to the ‘My Records’ tab in your CITI account to access the completion report/certificate.
IMPORTANT NOTES on COURSE SUBSTITUTIONS
CRAs / PIsIn special circumstances, Clinical Research Coordinators and Principal Investigators
COMMUNITY PARTNERS - CIRTificationFor community members who are involved in community engagement research, CIRTification
is a training option that can be substituted for the CITI Biomedical Investigator
Course training.
|
CITI Additional Information
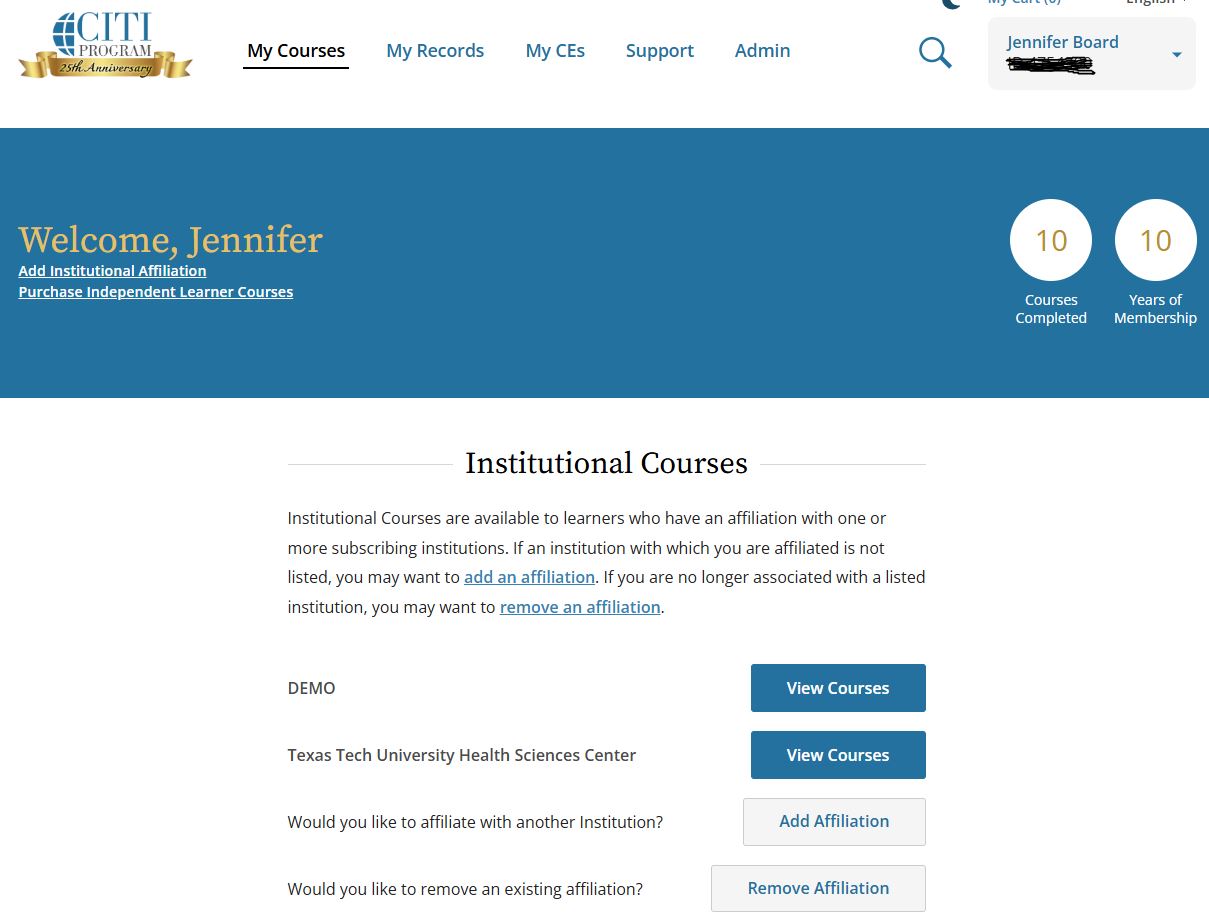
NOTE: If you have completed training at more than one institution, you can access all of them within one CITI account by adding an Institutional Affiliation for each.
- Log in to your CITI account
- Once logged in, the page should default to the ‘My Courses’ tab.
- Refer to the screenshot below, and you will see there are a few places to add an Institutional Affiliation. Click on one of the options, then type in the name of the institution.
 |
- Continue setting up your account by following the Registration steps #1-7 below.
- Steps 1-6: Answer the questions regarding ‘Personal Information’. NOTE: Register with your email for that institution. NOTE: If you are interested in CEU credits, make sure you answer the questions in Step 5 accordingly.
- Step 7: You will see a list of courses provided by the institution you are adding, noting the list will be different for each institution. Select the course(s) that are of interest and/or required. When you are done adding courses, click ‘Complete Registration’.
- Now when you go to your ‘My Courses’ tab, you will see all of your affiliated institutions listed under 'Institutional Courses'. Click on 'View Courses' next to the institution name, and you will see a list of the courses added for that institution.
NOTE: You can manage the courses listed in your CITI account as needed. If you do not see a course you need, you can add it. Likewise, if you have a course listed that you no longer need, you can remove it. You will still be able to access the course history under 'Course Completion History'.
- Log in to your CITI account
- Once logged in, the page should default to the ‘My Courses’ tab.
- Scroll down to the bottom of the page, past the list of your courses, and you will
see the last section titled, 'Learner Tools for Texas Tech University Health Sciences Center'. (Refer to the screenshot below)
- 'Add a Course': Click on this and you will see a complete list of all the courses provided by TTUHSC. Choose the course(s) to be added.
- 'Remove a Course': Click on this and you will see a complete list of your current courses. Choose
the course(s) to be removed.
- Once you have chosen the course(s) to be added/removed, scroll to the bottom and click 'Submit'.
- Click on the 'My Courses' tab to view your updated course list.
 |
- Log in to your CITI account
- Once logged in, the page should default to the ‘My Courses’ tab.
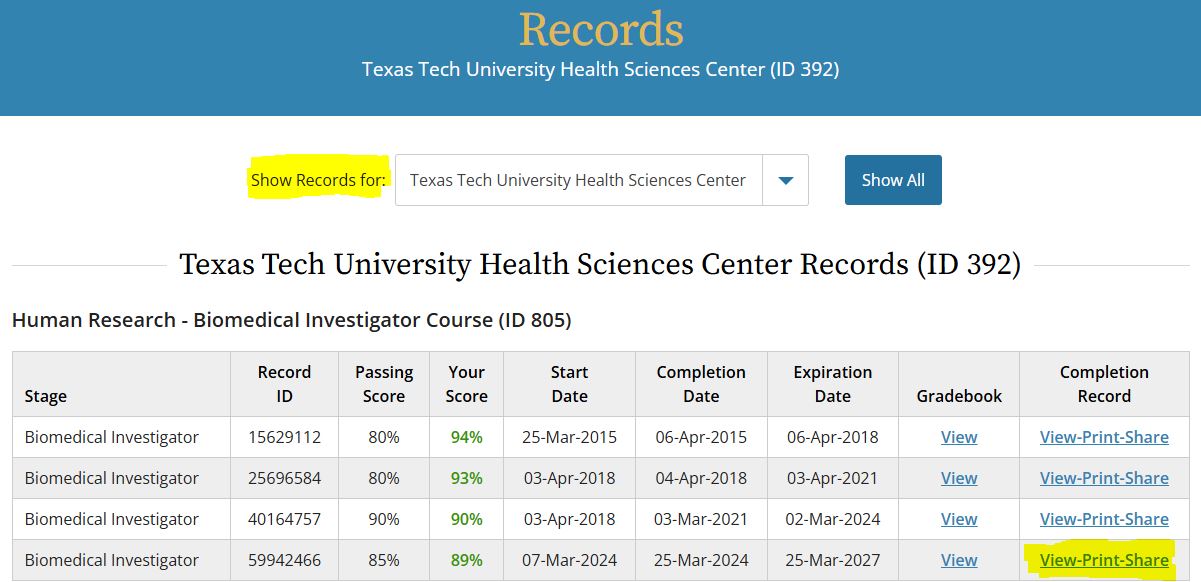
- Click on the 'My Records' tab to start. You will see a complete list of all the CITI courses you have completed.
- At the top of the 'My Records' page is a drop down titled 'Show Records for:'. If you are affiliated with more than one institution, you can look at each individually, or all of your institutions together. (Refer to the screenshots below)
- Scroll down until you find the course you need the completion certificate/report for.
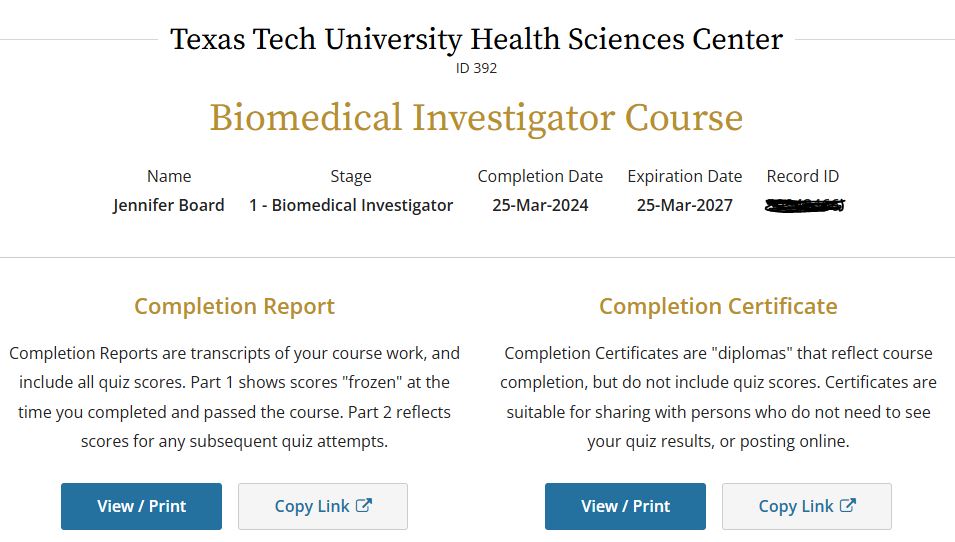
- Click on the 'View-Print-Share' link under 'Completion Record'.
- You will see two options - 'Completion Report' and 'Completion Certificate'. The report is the detailed version of the course, while the certificate is the overall view. The certificate is what most people ask for.
- Under each of these will be the option to 'View/Print' as well as 'Copy Link'. 'View/Print' is best for when you want to look at the certificate and save it. 'Copy Link' works well when you need to send the certificate to someone else.
 |
 |
TTUHSC offers several courses pertaining to Human Subject research, some of which may also be required by your department, funding agencies, and/or research sponsors. The courses are available at no charge to TTUHSC researchers through the TTUHSC’s CITI subscription.
These courses can be found under the ‘My Courses’ tab in your CITI account. (You can find instructions to add a course in the section above - 'Adding a course to your CITI account'.)
- Clinical Research Coordinator (CRC)
- Responsible Conduct Of Research (RCR)
- GCP for Clinical Trials with Investigational Drugs / Medical Devices (U.S. FDA Focus)
- Essentials of Public Health Research
- Revised Common Rule
- Institutional/Signatory Official: HSR
- Mental Health for Higher Ed and Healthcare
- COVID-19 series
- Research Security course
NOTE: The ONLY course REQUIRED by TTUHSC HRPP is the “Biomedical Investigator Course”.
Although TTUHSC offers several courses, there are many more that we do not have subscriptions to. You can still have access to most of these courses offered by CITI by signing up for them as an 'Independent Learner'.
You can find instructions and more information through this CITI link.
Additionally, you can find the list of available courses on your 'My Courses' tab. In the blue banner at the top of the page, under Welcome!, you will see 'Purchase Independent Learner Courses' listed. Clicking on that link will provide you with the current available courses,
as well as the cost of each. It will also include the list of available CITI Webinars
that you can sign up for (and their cost).
 |
Users have the option to purchase CE/CME credits for CITI courses they have completed.
The first step is to make sure that you have registered for CE credits, which is done under the ‘My CEs’ tab in your CITI account. Once registered, you can purchase credits for courses you have already completed AND/OR for courses you are getting ready to take. All of the key information you need, including instructions for payment, will show on the first page of each course. NOTE: Any cost associated is your responsibility.
CME credits ARE available for:
- Biomedical Investigator Training course
- Clinical Research Coordinator (CRC)
- Biomedical Responsible Conduct of Research (RCR)
- GCP for Clinical Trials with Investigational Drugs and Devices (U.S. FDA Focus)
- Revised Common Rule
CME credits ARE NOT available for:
- Essentials of Public Health Research
- Clinical Trial Billing Compliance (CTBC)
For any and all questions related to CE/CME credits, contact CITI Support
.
Click HERE for the PDF version of instructions on this page for CITI training. This PDF includes a complete list of the modules for the Biomedical Investigator Course (required and optional).

